Pharmaceutical Sieve Shaker, Doebritz Industrial Sifter, 316L Stainless Steel / ATEX / High-Throughput Powder Classification
Doebritz Pharmaceutical Sieve Shaker ensures zero cross-contamination and cGMP compliance. 316L stainless steel, ATEX certified, and high-containment design for API powders. OEM ready.
Product Summary
Engineered for sterile pharmaceutical powder classification, the Doebritz Pharmaceutical Sieve Shaker delivers high-throughput sieving from 5 kg/h to 5,000 kg/h with a separation range of 25µm to 5,000µm. Constructed entirely from AISI 316L stainless steel with an electropolished finish (Ra ≤ 0.4µm), this unit is fully ATEX II 2D Ex h IIIC T120°C Db certified for safe operation in hazardous dust environments. Capable of processing hygroscopic powders, granules, and fragile tablets without product degradation.
Why Source Doebritz Pharmaceutical Sieve Shaker
- Zero Cross-Contamination Guarantee: Fully crevice-free welding, sanitary tri-clamp connections, and quick-release sieve frames enable complete wash-down and validation per cGMP and FDA 21 CFR Part 11.
- Superior Screening Efficiency: Unique eccentric-weighted drive system generates adjustable 3D elliptical vibration, eliminating screen blinding and achieving >99.5% separation accuracy for API powders.
- Global OEM Compliance: All Doebritz units meet USP , EP 2.9.38, and ISO 18062 particle contamination standards. Full documentation packages including IQ/OQ/PQ protocols available.
- 30% Longer Sieve Life: Patented spring-mounted suspension absorbs mechanical shock, reducing mesh fatigue and tearing compared to conventional gyratory sifters.

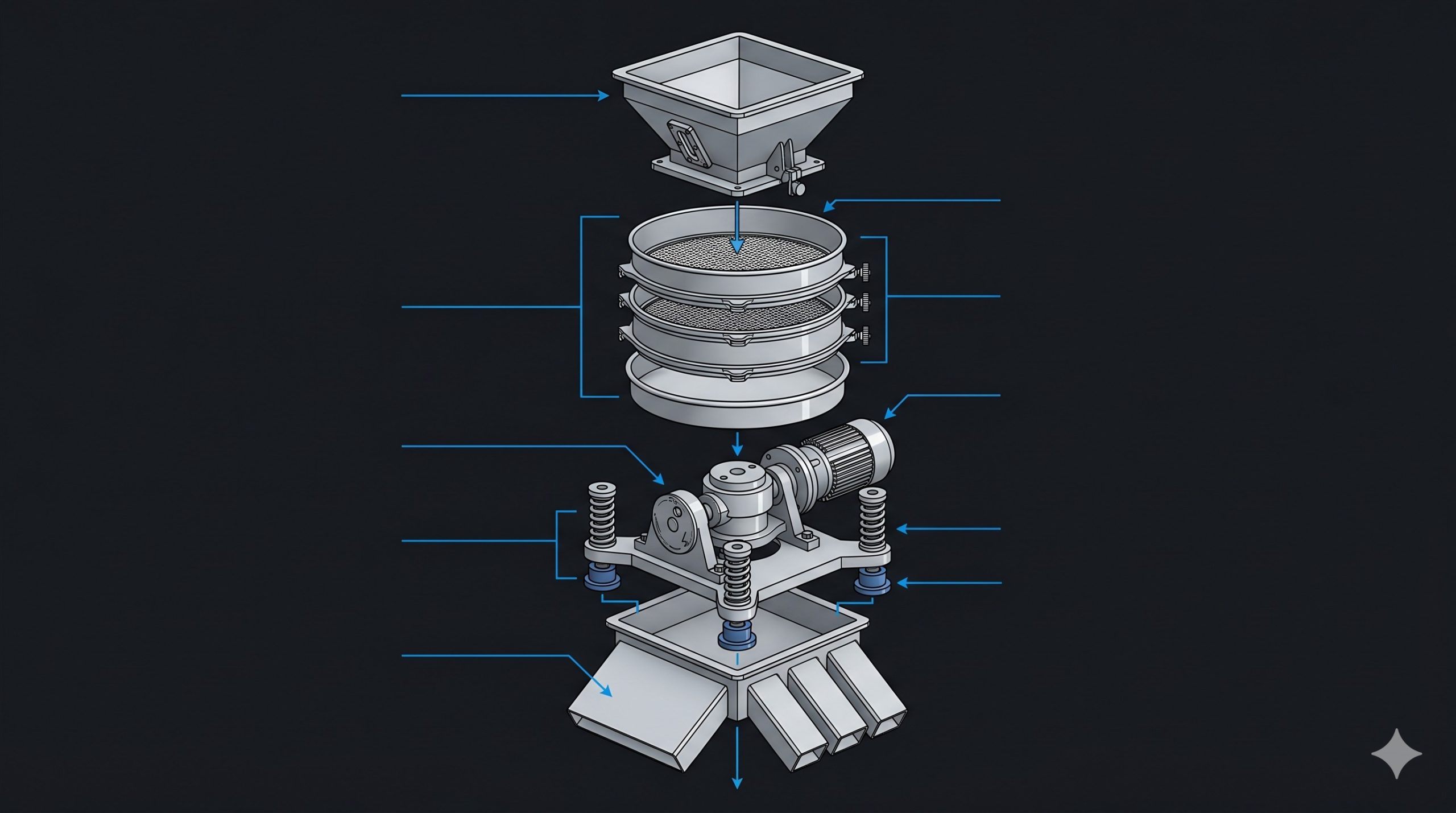
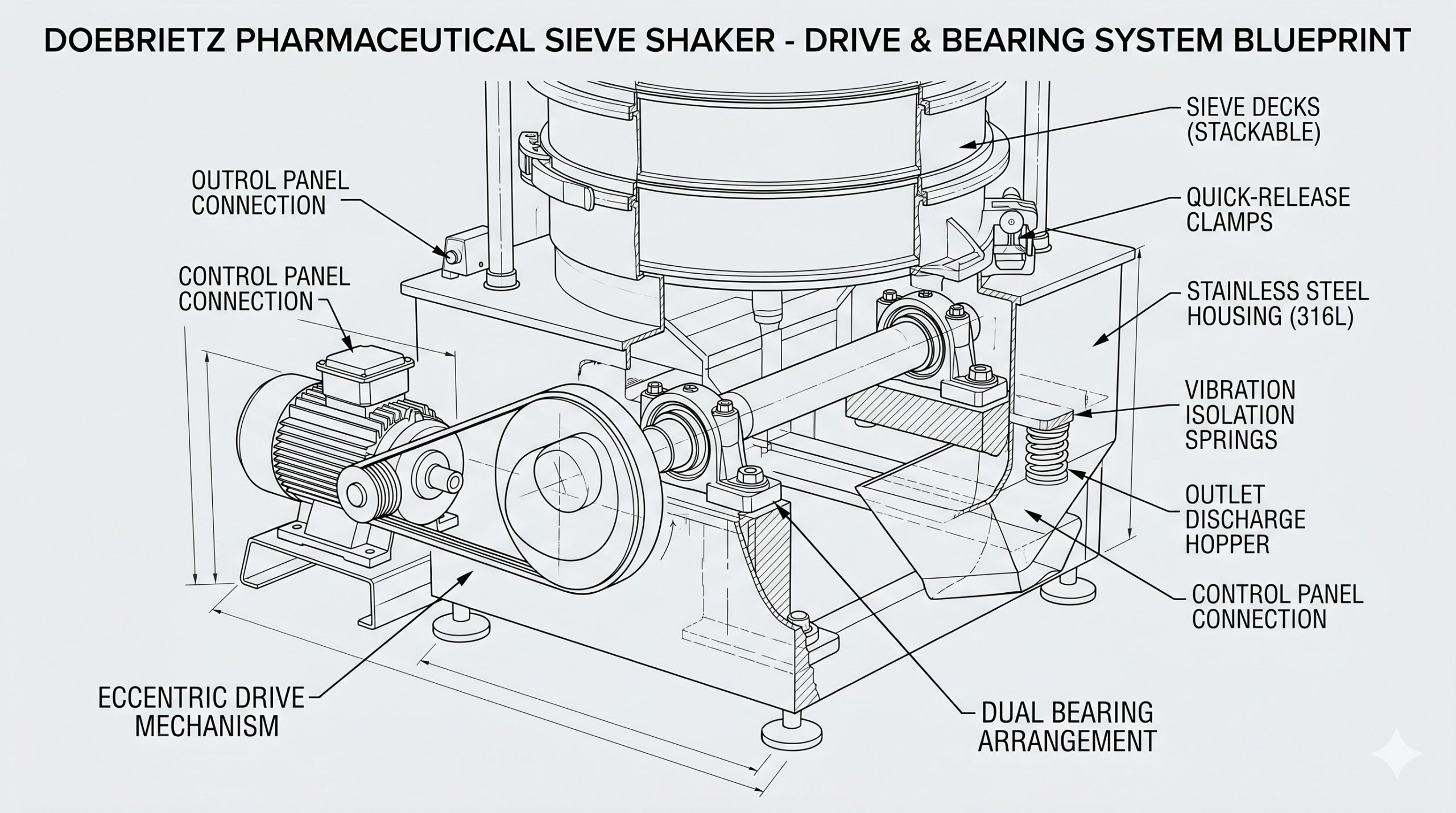
Housing & Rotor Engineering
The Doebritz Pharmaceutical Sieve Shaker features a monobloc housing precision-cast from 316L stainless steel with no internal welds or dead zones where product could accumulate. The eccentric drive shaft is supported by dual SKF Explorer sealed-for-life bearings with IP69K-rated labyrinth seals, preventing lubricant migration into the product zone. The sieve frame clamping system utilizes a single over-center lever instead of multiple bolts, reducing screen changeover time from 30 minutes to under 3 minutes. All contact parts are interchangeable with standard 450mm or 600mm diameter pharmaceutical-grade test sieves.
Technical Specifications
Each Doebritz unit undergoes 100% factory acceptance testing (FAT) with traceable documentation. Parameters below represent the standard PS-600 model; custom sizing and explosion-proof motors available upon request for global OEM supply chains.
| Specification | Detail |
|---|---|
| Body Material | AISI 316L Stainless Steel (Electropolished, Ra ≤ 0.4µm) |
| Rotor / Drive Design | Eccentric-weighted horizontal shaft with dual SKF bearings |
| Sieve Diameter | 450 mm or 600 mm (standard pharmaceutical test sieve compatible) |
| Separation Range | 25 µm to 5,000 µm (80 mesh to 500 mesh) |
| Throughput (Powder) | Up to 5,000 kg/h (depending on bulk density and mesh size) |
| Motor Rating | 1.1 kW – 3.0 kW, 230/400V, 50/60Hz, IP66 |
| Ex Certification | ATEX II 2D Ex h IIIC T120°C Db |
| Operating Temp Range | -20°C to +150°C (with high-temp silicone gaskets) |
| Sealing Type | Silicone / FKM / EPDM (FDA-compliant, crevice-free) |
| Surface Finish | Mirror polish (Ra ≤ 0.4µm) internal & external |
Heavy-Duty Application Scenarios
Oral Solid Dose (OSD) Powder Pre-Sieving
Installed upstream of tablet press feed frames, the Doebritz shaker removes agglomerates and foreign particles from excipients like lactose, MCC, and magnesium stearate, ensuring uniform die fill and weight consistency.
High-Potency API (HPAPI) Containment
Configured with split butterfly valves and continuous liner systems, the shaker achieves <1µg/m³ occupational exposure levels (OEL) for Category 4 and 5 compounds. Available with passivation certification and gamma irradiation-ready sieves.
Frequently Asked Questions
- What is the minimum order quantity (MOQ) for Doebritz pharmaceutical sieve shakers?
- Doebritz offers flexible MOQs: 1 unit for lab-scale R&D (PS-200), 5 units for commercial production lines. For global OEM contract manufacturing, we support consignment stock programs with 2-week lead times after initial batch.
- Can you customize the sieve shaker for non-standard pharmaceutical powders like lyophilized cakes or sticky APIs?
- Yes. Our engineering team integrates ultrasonic anti-blinding systems for sub-45µm sieving of cohesive APIs, plus Teflon-coated mesh or nylon screens for hygroscopic or electrostatic powders. Provide your material characterization report (PSD, angle of repose, moisture content) for a free engineering review.
- Is the Doebritz unit compliant with ATEX and what documentation is included?
- All Doebritz Pharmaceutical Sieve Shakers carry ATEX II 2D Ex h IIIC T120°C Db certification (zone 22 interior, zone 21 exterior). Deliverables include: EC type examination certificate, declaration of conformity, user manual with hazardous area installation drawings, and certified drawings for system integrators.
- What is the delivery lead time for an FDA/USP-compliant unit to North America or Europe?
- Standard lead time is 6-8 weeks for ATEX-certified 316L models, including FAT at our German facility. For urgent OEM projects, accelerated 4-week delivery is available with air freight. All units ship with sterile-wrapped contact parts and passivation certificates.
- Does Doebritz provide validation support or spare parts globally?
- Absolutely. We supply full IQ/OQ/PQ protocol templates, factory calibration certificates (traceable to PTB), and a 2-year warranty on all non-wearing components. Spare parts (sieve frames, mesh, springs, bearings) are stocked in our US, EU, and Singapore warehouses for 48-hour emergency dispatch.






Reviews
There are no reviews yet.